The field of metabolic disease treatment is ushering in a storm of change.
GLP-1, a target once confined to diabetes treatment, has now emerged as a "superstar" in the global pharmaceutical industry. According to Evaluate Pharma's forecast, the market size of GLP-1RA will exceed 130 billion US dollars by 2030.
Amid the market enthusiasm, in addition to its widely concerned potential for multi-field applications—some people are concerned about whether it can "protect the brain", improve mood, or even help reduce the "craving" for alcohol or nicotine—on the other hand, enterprises focusing on the R&D and production of pharmaceutical peptides are also accelerating their layout to fully seize the high-quality GLP-1 track.
Molecular Biology and Signal Transduction of GLP-1R
Glucagon-like peptide-1 receptor (GLP-1R) belongs to the class B G protein-coupled receptors, which are mainly expressed in pancreatic β cells, cardiomyocytes, vascular endothelium and kidneys. After ligand binding, it enhances glucose-dependent insulin secretion through the Gαs/cAMP/PKA pathway, and simultaneously activates PI3K-Akt and ERK1/2 signals to exert anti-inflammatory, antioxidant and endothelial protective effects.
1. SUSTAIN-6 Study: Beyond Hypoglycemia, Significant Reduction in Cardiovascular Risk
The SUSTAIN-6 study, published in 2016, was the first RCT to confirm the benefit of GLP-1R agonists on hard cardiovascular endpoints. This study was a CVOT of semaglutide 0.5/1.0 mg injection, aiming to explore the impact of semaglutide 0.5/1.0 mg injection or placebo on the incidence of MACE in T2DM patients with CVD/high risk on the basis of standard treatment. A total of 3,297 T2DM patients with high cardiovascular risk were enrolled and randomly assigned to receive weekly subcutaneous injection of semaglutide 0.5 mg, 1.0 mg or placebo, followed up for 2 years. The results showed that the risk of MACE (cardiovascular death, non-fatal myocardial infarction, non-fatal stroke) was reduced by 26% (HR 0.74; 95%CI 0.58–0.95), and the main benefit came from the reduction of non-fatal myocardial infarction.
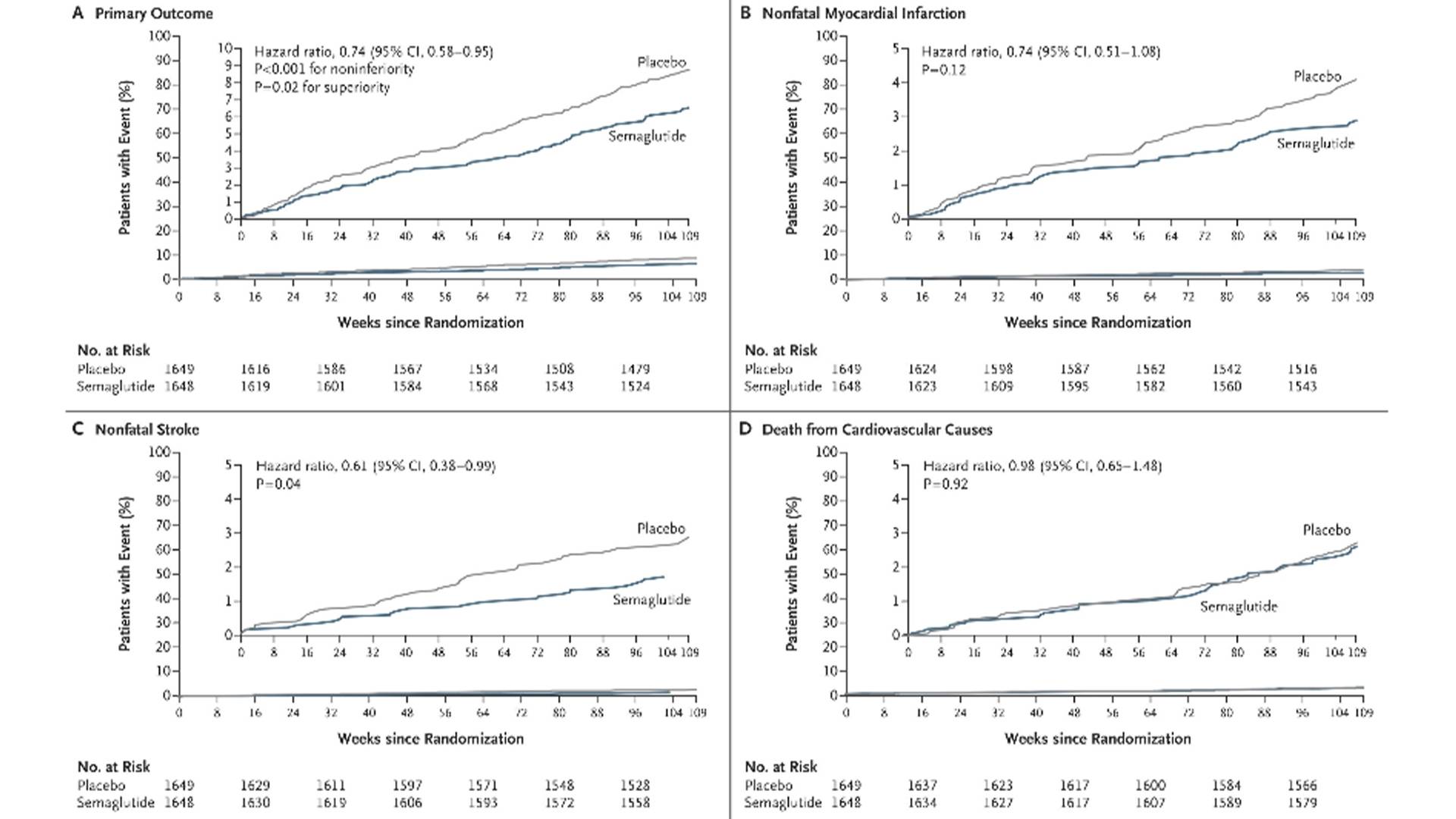
Cardiovascular Outcomes
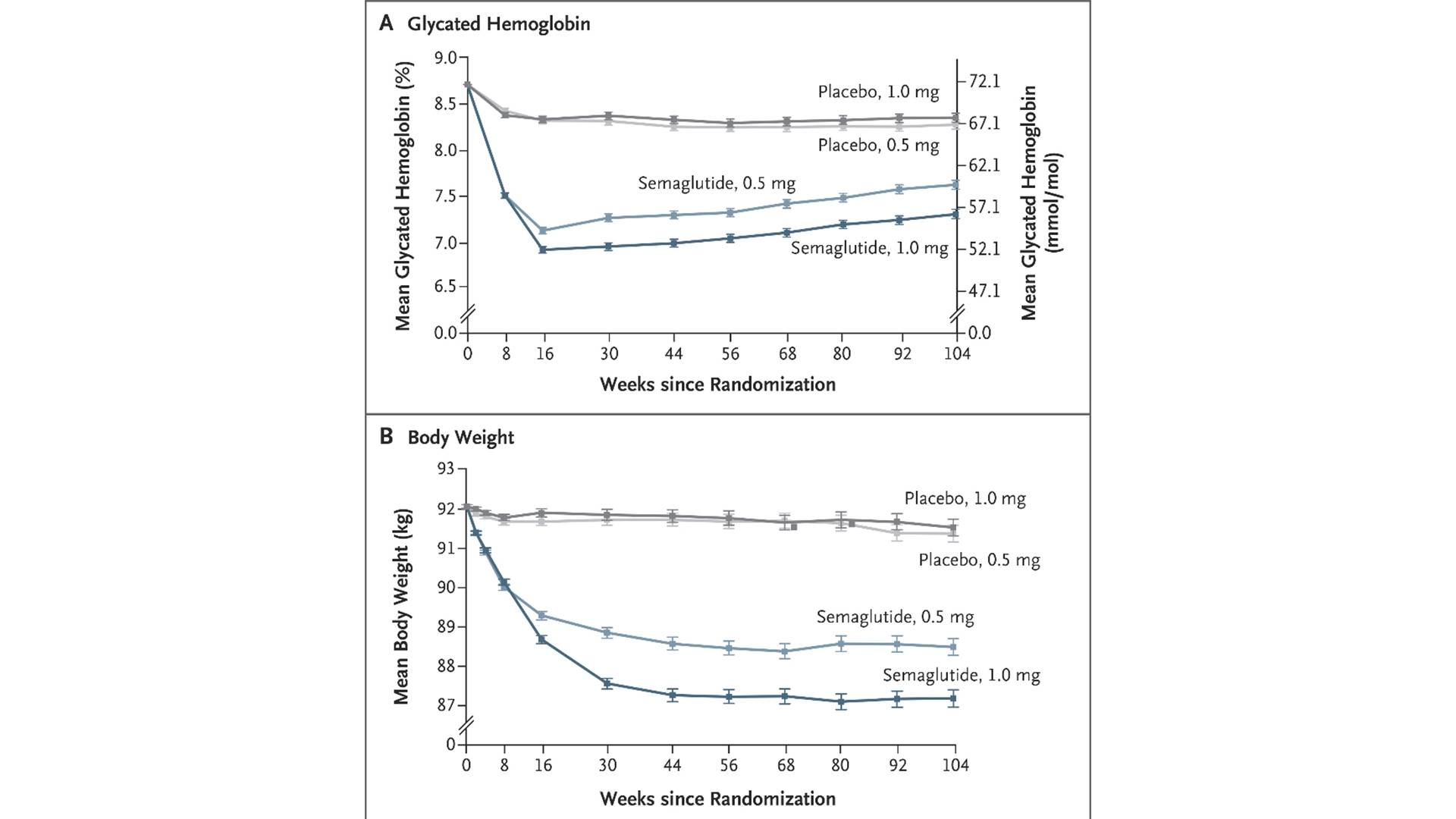
Glycated Hemoglobin and Body Weight
2. SELECT Trial: Primary Cardiovascular Prevention in Non-Diabetic Populations
In 2023, the SELECT trial, published in NEJM, enrolled 17,604 overweight/obese (BMI≥27kg/m²) patients diagnosed with ASCVD but without diabetes, who were randomly assigned to semaglutide 2.4 mg once a week or placebo, with a mean follow-up of 39.8 months. The results showed that the risk of MACE was reduced by 20% (HR 0.80; 95%CI 0.72–0.90), cardiovascular mortality decreased, and the benefit was independent of the magnitude of weight loss. Post-hoc analysis suggested that a 3.3 mmHg reduction in systolic blood pressure and a 37.8% decrease in hs-CRP partially explained the event curve separation that occurred early (<6 months).
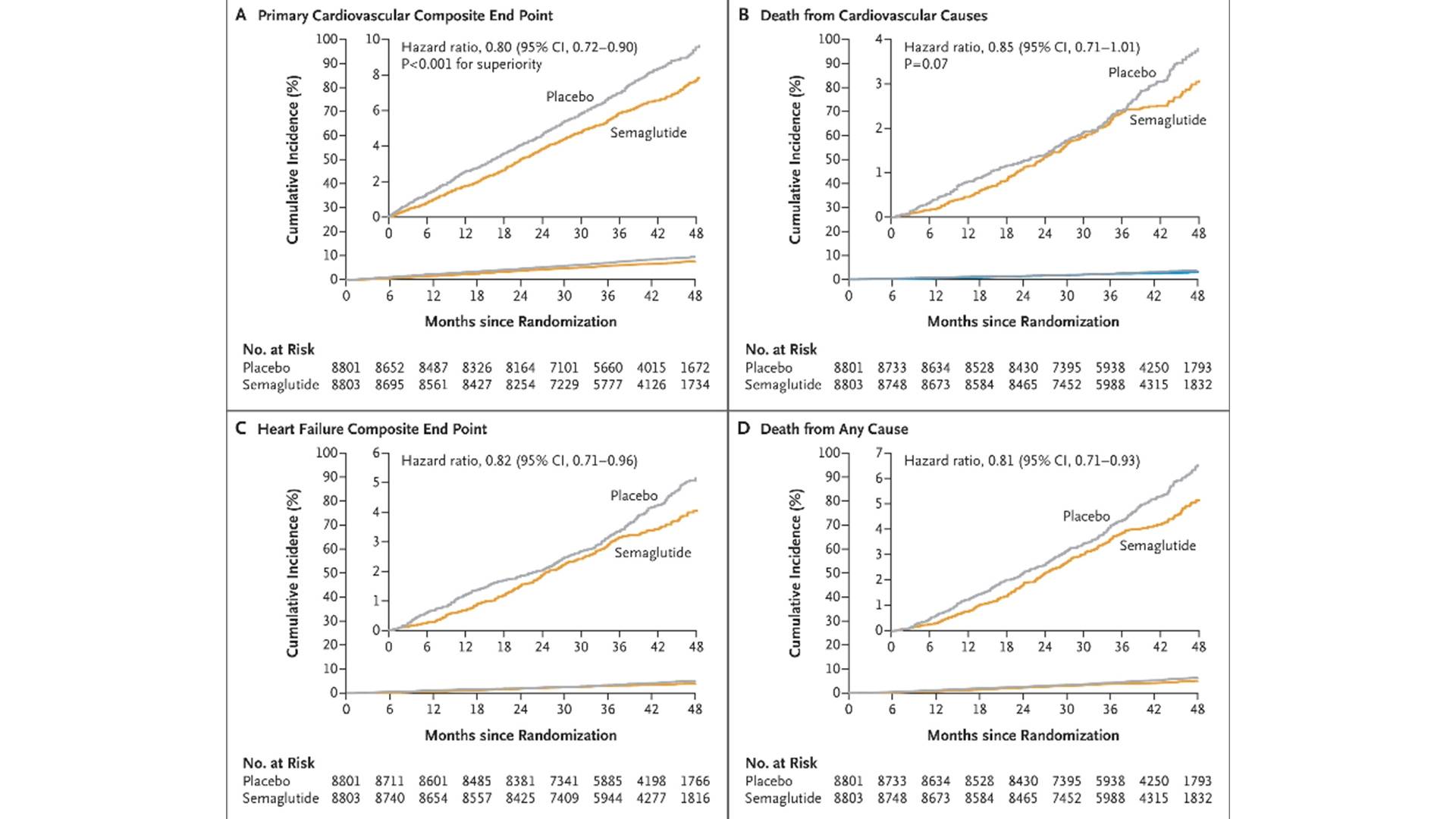
Time to First Occurrence of Primary and Secondary Endpoints
3. SOUL Study: Real-World Clinical Practice
The SOUL study, presented at ACC 2025, was an international multi-center, randomized, double-blind, parallel-group, placebo-controlled phase Ⅲb CVOT. The study covered 444 research centers in 33 countries and regions around the world, enrolling a total of 9,650 T2DM patients with ASCVD and/or CKD, who were randomly assigned to the oral semaglutide tablet group (4,825 cases) and the placebo group (4,825 cases). The results showed that 668 cases (13.8%) of MACE occurred in the placebo group, while 579 cases (12.0%) occurred in the oral semaglutide tablet group, with a relative risk reduction of 14% (HR 0.86; 95%CI: 0.77~0.96, P=0.006), and the superiority statistical hypothesis of the primary endpoint was achieved (Figure 2). The cardiovascular benefits of oral semaglutide tablets were consistent across subgroup analyses of different genders, ages, BMI, eGFR, and concomitant medications.
4. STRIDE Trial: Improving Motor Function in Patients with Symptomatic PAD
The STRIDE trial was the first large-scale RCT designed specifically for improving the function of T2DM patients with symptomatic peripheral arterial disease (PAD) in the field of hypoglycemic drugs. This was a multinational multi-center, randomized, double-blind, placebo-controlled, phase 3 clinical trial, enrolling 792 T2DM patients with intermittent claudication, aiming to evaluate whether adding 1.0 mg semaglutide injection on the basis of standard treatment can improve the exercise capacity of T2DM patients with symptomatic PAD. The results showed that after 52 weeks, compared with placebo, semaglutide increased the maximum walking distance (MWD) on a constant-load treadmill (3.2km/h, 12% slope) by 13% (P=0.0004), with a mean increase of 39.9m and a median increase of 26.4m; at the same time, the pain-free walking distance (PFWD) was significantly increased by 11% (P=0.0046), with a mean increase of 29.8m and a median increase of 13.8m.
GLP-1R/GIPR Dual Agonists: The "Metabolic-Cardiovascular" Dual Advantages of Tirzepatide
GIPR and GLP-1R are co-expressed in pancreatic islets and adipose tissue, and dual agonism can synergistically enhance insulin sensitivity, promote white adipose browning and improve lipid oxidation.
The SUMMIT study , presented at the 2024 AHA Annual Meeting, was an international, double-blind, randomized, placebo-controlled trial aiming to observe the impact of tirzepatide on the risk of heart failure events and quality of life in obese patients with HFpEF. The study enrolled 713 heart failure patients from 9 countries/regions, with an ejection fraction ≥50% and BMI ≥30 kg/m². Patients were randomly assigned to the tirzepatide group or the placebo group in a 1:1 ratio, receiving tirzepatide (up to 15 mg, subcutaneous injection, once a week) or placebo for at least 52 weeks. The results showed that compared with placebo, tirzepatide significantly reduced the risk of worsening heart failure events in patients with HFpEF combined with obesity, and also significantly improved the patients' heart failure symptoms and physical conditions.
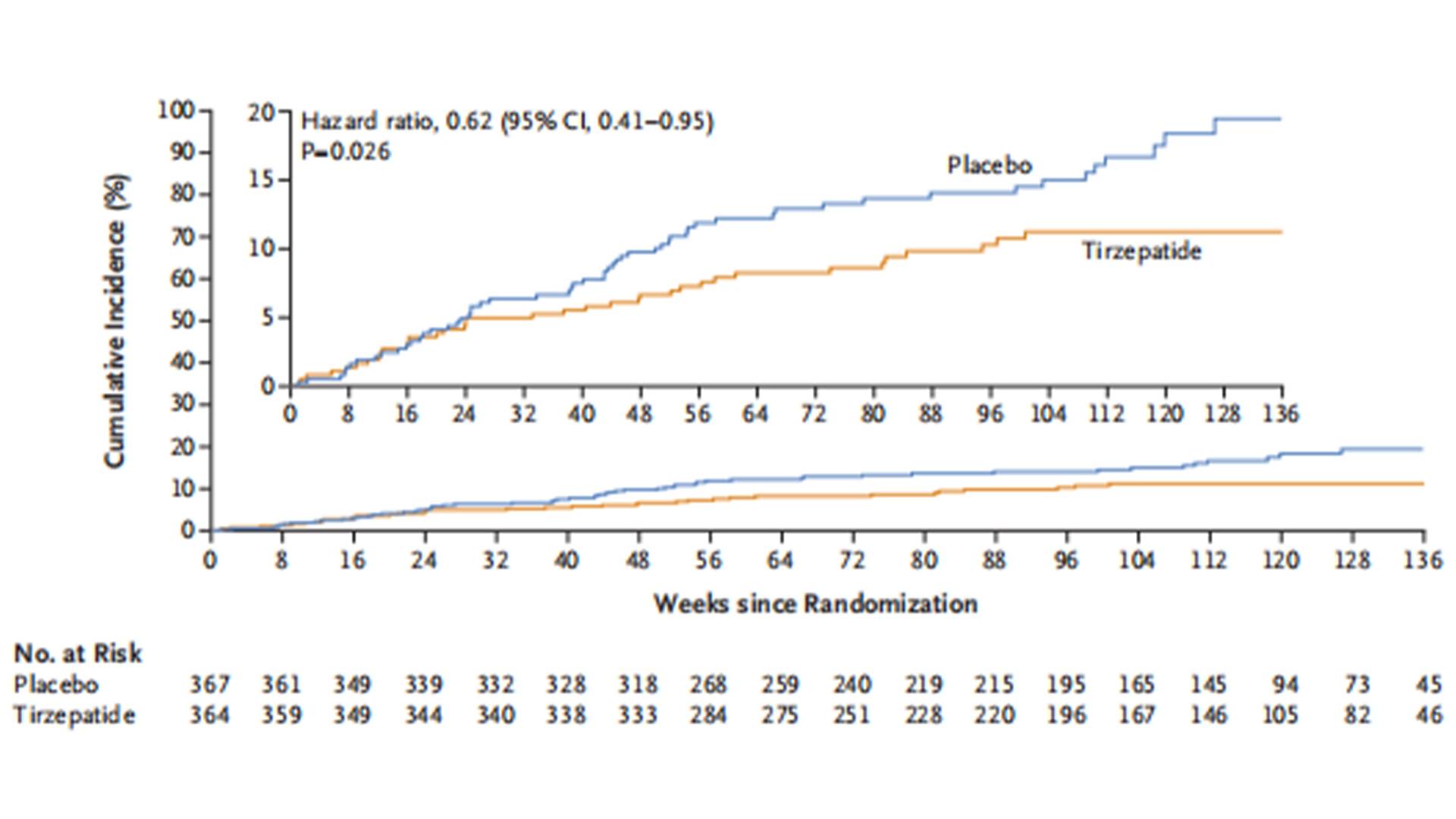
Composite Events of Death Due to Cardiovascular Disease or Worsening Heart Failure
Safety, Tolerability and Key Points for Clinical Implementation
The most common adverse events of GLP-1R agonists are mild to moderate gastrointestinal reactions (nausea, vomiting, diarrhea), which are dose-dependent and usually resolve spontaneously within 4–8 weeks. Neither SELECT nor meta-analyses observed an increased risk of pancreatitis, medullary thyroid carcinoma or severe infection, but the discontinuation rate of semaglutide treatment was higher than that of placebo (RR 1.67; 95%CI 1.27–2.21), suggesting that clinical practice needs to strengthen patient education and dose titration management. For T2DM patients with ASCVD or high-risk factors, the 2025 ADA Guidelines recommend selecting GLP-1R agonists with proven cardiovascular benefits; for obese patients without diabetes but diagnosed with ASCVD, semaglutide 2.4 mg/week has been approved by the FDA for the dual indications of "weight loss and cardiovascular risk reduction".
Conclusion
GLP-1R has gradually emerged as a versatile target spanning glucose metabolism, weight management, atherosclerosis and heart failure. Landmark trials such as SUMMIT and SELECT have proved that both the single agonist semaglutide and the dual agonist tirzepatide can significantly reduce MACE and reshape the management model of metabolic chronic diseases. In the future, precise typing, combined treatment and real-world evidence around GLP-1R will further consolidate its position, making GLP-1R agonists a model of "one drug for multiple targets" and redefining the comprehensive management of metabolic chronic diseases.

Industrial development is based on raw materials. The continuous heating up of the GLP-1 track has put forward strict requirements on the purity, stability and safety of upstream pharmaceutical peptide raw materials—peptide synthesis process is complex, impurity control is difficult, and purity directly determines the biological activity and clinical safety of raw materials. The conventional industry reference purity standard is ≥96.0%, while high-purity raw materials are even scarcer resources. Against this background, Xi’an Jenifer Technology Co., Ltd. has stood out in the GLP-1 raw material field with its profound accumulation in the R&D and production of bioactive ingredients. The GLP-1 produced by the company has a purity of up to 98%, far exceeding the conventional industry standard. It not only maximizes the retention of its biological activity, but also maintains a high homology with human endogenous GLP-1 (7-36), ensuring the precise exertion of its effects in blood glucose regulation, weight loss and cardiovascular protection; furthermore, by optimizing the synthesis process and purification technology, it effectively controls impurity content and solvent residue, reduces potential risks in clinical application, and solves the industry pain points of insufficient purity and unstable activity of traditional GLP-1 raw materials.

